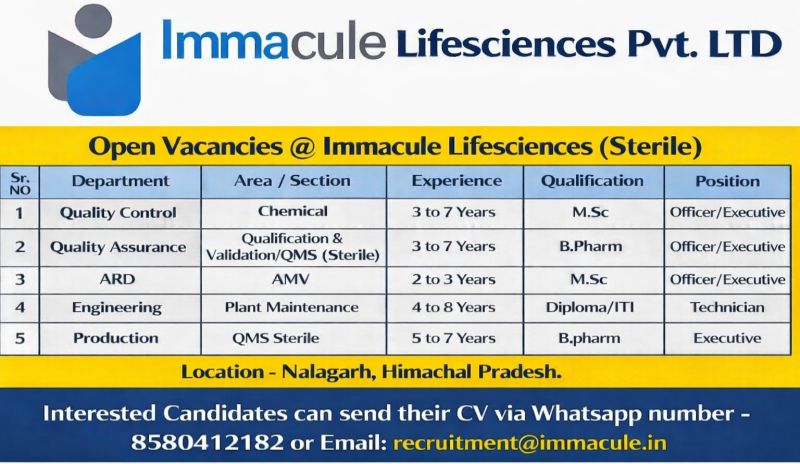
Company Introduction
Immacule Lifesciences Pvt LTD operates sterile pharmaceutical facilities supported by structured documentation and regulatory aligned quality systems.
Core Responsibilities
- Verify completed batch records against approved master documents on the shop floor.
- File reviewed documents into controlled archives using indexed folders.
- Inspect cleanroom areas using printed checklist sheets and sign inspection records.
- Issue controlled formats, registers, and labels from document control cabinets.
- Check training records and file signed attendance sheets.
- Maintain logbooks for document issuance and retrieval.
Skills and Technical Exposure
- Batch manufacturing records and master formula files.
- Good manufacturing practice manuals.
- Controlled document registers and logbooks.
- Training matrices and qualification files.
- Inspection checklists and review forms.
Experience Context
- Work involves routine presence inside sterile manufacturing plant areas.
- Daily document volumes cover multiple sterile batches and supporting records.
- Without this role, batch release readiness and audit preparation fail.
How to Apply
recruitment@immacule.in