Company Introduction
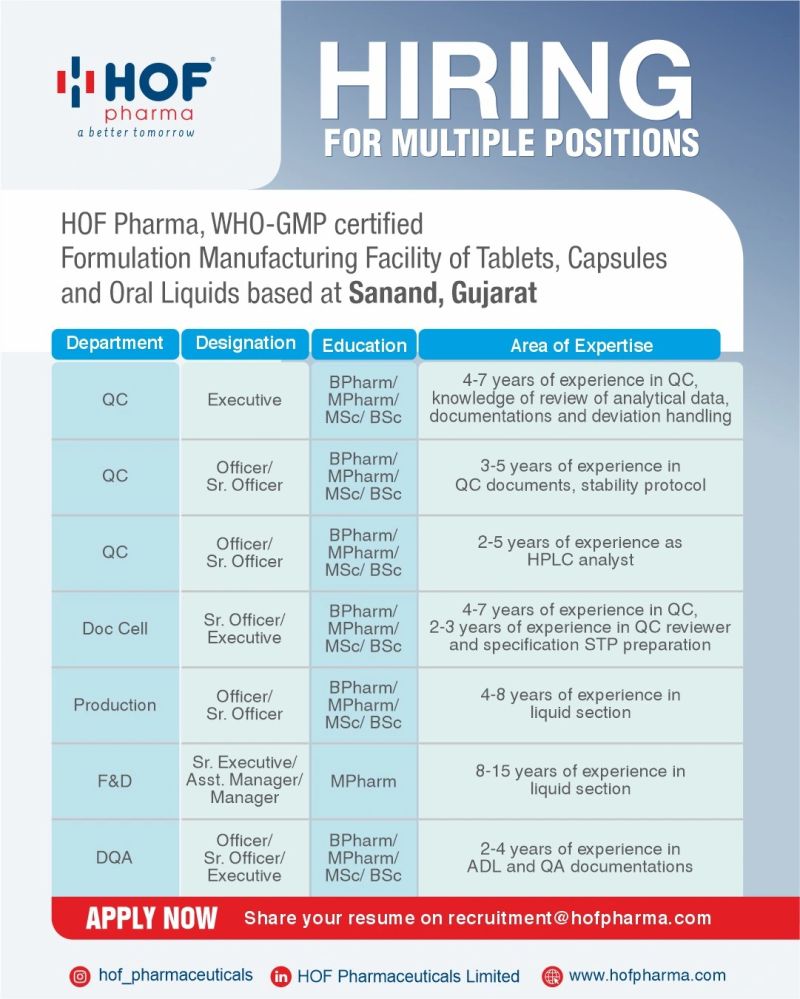
HOF Pharma operates a WHO-GMP certified formulation facility manufacturing tablets, capsules, and oral liquid products for regulated markets.
Core Responsibilities
- Perform sample analysis using calibrated HPLC systems and documented test methods.
- Record raw data in laboratory notebooks and approved analytical worksheets.
- Verify assay, dissolution, and impurity results against registered specifications.
- Prepare stability sample labels, storage logs, and pull schedules.
- Review chromatograms, system suitability records, and calculation sheets.
- Maintain reference standards, working standards, and reagent inventory logs.
Skills & Technical Exposure
- HPLC and UV-visible spectrophotometer instrumentation.
- Analytical test methods for finished dosage forms.
- Laboratory notebooks and controlled analytical worksheets.
- Stability protocols and sample storage registers.
- Chromatography data systems and integration software.
Experience Context
- Work takes place inside a GMP quality control laboratory within a formulation plant.
- Daily testing volumes cover multiple commercial and stability batches.
- Without this role, product release timelines stall due to incomplete analytical evidence.
How to Apply
recruitment@hofpharma.com