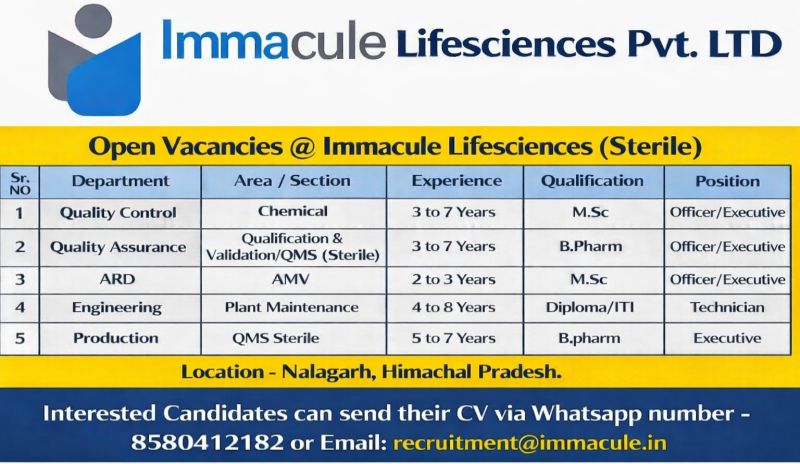
Company Introduction
Immacule Lifesciences Pvt LTD manufactures sterile pharmaceutical products using validated production systems and controlled environments.
Core Responsibilities
- Prepare production areas by checking equipment status and line clearance sheets.
- Operate sterile manufacturing machines following approved batch records.
- Record production data in batch documents during filling activities.
- Clean production equipment using defined cleaning records.
- Label and transfer finished units to designated storage areas.
Skills and Technical Exposure
- Sterile production equipment.
- Batch manufacturing records.
- Line clearance documents.
- Cleaning records and checklists.
- Controlled environment monitoring tools.
Experience Context
- Work occurs inside sterile pharmaceutical production suites.
- Production volumes include multiple sterile batches per shift.
- Without this role, filling schedules stop and batch output reduces.
How to Apply
recruitment@immacule.in